THE LANCET
Lancet July 1, 1995; Vol 346: 29-32
Public HealthScreening mammography and public health policy: the need for perspective
Charles J Wright, C Barber Mueller
Summary
The early trials of screening mammography, reporting 30% relative reduction in mortality from breast cancer in women over 50 years of age, led to strong professional and public demand for screening programmes. There has been little publicity about the subsequent trials showing no significant benefit in any age group, or about the harm and costs associated with screening mammography. For women under 50, there is a reluctant consensus that screening is not beneficial, but there is increasing pressure for publicly funded programmes for older women.
When analysed in terms of population benefit, the randomised controlled prospective trials showed that the numbers of women screened to achieve one less death per year ranged ffom 7086 (Health Insurance Plan of New York), to 63264 (Malmo), to infinity (Canadian National Breast Screening Study). About 5% of screening mammograms are positive or suspicious, and of these 80-93% are false positives that cause much unnecessary anxiety and further procedures including surgery. False reassurance by negative mammography occurs in 10-15% of women with breast cancer that will manifest clinically within a year. Our calculations confirm others that the mean annual cost per life "saved" is around $1.2 million (£558 000).
In the allocation of limited resources, public health policy on a proposed mass population intervention must be based on a critical analysis of benefits, harm, and cost. Since the benefit achieved is marginal, the harm caused is substantial, and the costs incurred are enormous, we suggest that public funding for breast cancer screening in any age group is not justifiable.
Follow-up studies show that breast cancer is a devastating disease, with an annual death rate of around 8% among survivors even 20 years after diagnosis.[1] These grim facts, coupled with the widespread feeling that small equals early, have led many people to think that the best hope for improving results must lie with earlier diagnosis. Mammography is certainly capable of identifying abnormalities that may be breast cancer at a clinically undetectable stage. The Breast Cancer Detection Demonstration Project (BCDDP)[2] showed a 0.5% incidence of breast cancer among over 250 000 women screened; many of these lesions would not have become clinically apparent until some unknown time later.
However, the more important question is whether early putative diagnosis benefits patients in terms of overall longevity. The problems of lead-time bias and length bias require evidence of a change in population mortality rather than an improvement in case-survival rates, which may occur without patient benefit as a result of earlier diagnosis. Outcomes such as the proportion of node-positive patients diagnosed or tumour size are of interest only insofar as they reflect on eventual mortality or choice of therapy. For public health policy decisions about allocation of resources for breast screening, one needs a measure of added years of life (or "lives saved") rather than lives "at risk" after diagnosis.
Researchers understandably present the data in the form that best illustrates their claim to benefits of the intervention they have been studying. With mammographic screening for breast cancer, results from the two oldest trials in women aged 50 years and more have been portrayed as a 30% reduction in mortality from the disease.[3-5] This figure has been translated into remarkably optimistic messages:
- "Most women with breast cancer could be saved by detection . . . with mammography"[6]
- "Mammography helps your doctor see breast cancer before there is a lump when the cure rates are near 100%" (American Cancer Society)[7]
- "If you are over 35 and haven't had a mammogram, you need more than your breasts examined" (television and magazine advertisements). Skrabanek used to call this kind of grossly inaccurate overstatement "wishful thinking".[8]
Statements such as these have raised public awareness, and with it has come an increasing clamour for mammography among women of all ages. Meanwhile, pleas for a more cautious approach based on comprehensive statistical and epidemiological analyses[5, 9] have largely been ignored. Three important and disturbing issues that arise from the evidence of published trials have so far received scant attention: (a) the diminishing return from the trials in historical sequence; (b) the size of benefit in relation to the harm caused by screening; and (c) the cost.
|
|
||||||
|---|---|---|---|---|---|---|
| HIP 1982 | SNBH 1985 | Malmo 1988 | Edin 1990 | Stockholm 1991 | CNBSS 1992 | |
|
|
|
|
|
|
|
|
| No of women screened | 31 881 | 78 085 | 21 088 | 23 226 | 40 318 | 44 854 |
| Deaths from breast cancer | ||||||
| Screened | 147 | 87 | 63 | 68 | 39 | 76¤ |
| Control | 192 | 127* | 66 | 76 | 58* | 67 |
| Reduction in mortality (relative %) | 23 (p<0.05) | 31 (p<0.02) | 5 (NS) | 11 (NS) | 33 (NS) | · · (NS) |
| No of women screened for 1 less death/year | 7086 | 13 665 | 63 264 | 20 322 | 15 703 | · · |
|
|
||||||
Table: Screening mammography: randomised prospective controlled clinical trials
Findings in sequential studiesThe results of six randomised prospective controlled clinical trials[4, 10-15] in chronological order of publication are summarised in the table. No study showed a significant benefit for women under the age of 50. The two early studies[4, 10] showed a significant relative reduction in mortality from breast cancer of 23-31 % (the absolute mortality reduction was 0.05-0.14 % of women screened), whereas the four most recent studies showed no significant benefit at any age. Further long-term reports from these studies are awaited. Meta-analyses of the later trials have shown a statistically significant reduction of deaths due to breast cancer, but only in women over 50.[16, 17]
How can one explain the apparently diminishing return in historical sequence? The increasing use of adjuvant chemotherapy may have been a factor, but is unlikely to be a confounder since it would appear in both arms of the trials. A noteworthy finding is that mortality falls sequentially even in the control groups in the separate trials but, by definition, single arms of randomised controlled trials cannot be compared against each other. Should the early trials be accepted as the gold standard and the later ones dismissed as somehow incompetent? Surely not, in view of the increasing rigour of trial design over the past 30 years and the vast improvement in quality of mammography. The later trials are probably a more accurate reflection of the limited value of screening. So why do people still advocate the screening of women under age 50? Until new evidence becomes available, the issue of screening women in that age group does not warrant further discussion. Moreover, the claimed extent of benefit in women over age 50 must be set against the harm induced by screening and the cost.
Fragile case for benefit
It is enlightening to analyse the trial results in terms of population benefit rather than relative mortality reduction. By way of illustration we will examine the results of the 1985 SNBH study, which showed a significant 30 % reduction in breast cancer mortality but no reduction in overall mortality. The figure illustrates the results per 10 000 women screened, based on this "best case" scenario. With the usual "positive call" rate of 5-10 %, at least 1500 women would have a positive mammogram if mammography were repeated regularly over 7 years, and 137 (1.37%) would have breast cancer diagnosed. There would be 11 (0.11%) deaths from the disease compared with 15 (0.15%) among 10 000 unscreened controls during the period of the study. Although these figures represent a 30 % reduction in mortality (relative risk reduction), the end result is that 4 women out of 10 000 screened would have benefited. The table shows the number of women who must be screened in each study for 1 less death from breast cancer per year: the range is 7086 (HIP) to 63 264 (Malmo). The CNBSS actually found a small increase in breast cancer mortally in the screened population, mainly due to the results in the 40-49 year age group. Deaths from colon cancer were reduced in the CNBSS screened group by almost the same number as the breast cancer deaths were increased.[2] As in all studies on screening mammography there was no difference in all-cause mortality between the screened and unscreened groups.
In view of what we know about the cytokinetics of the disease, we should not be surprised that the eventual outcome (death due to breast cancer) for the large majority of women is unaffected by screening mammography. The growth rates of breast cancers are highly variable, but in most cases the disease has been present for many years before it is diagnosable by any means. About 40 doublings of breast cancer cells create a lethal tumour burden, yet mammography cannot detect a mass until 25-30 doublings have already occurred.[18, 19]
Harm caused
Assessment of benefit is contentious, but assessment of harm caused by mammography is direct and simple: false-positives cause unnecessary interventions; false negatives give inappropriate reassurance. There are false hopes and expectations of cure, high levels of public fear and anxiety about breast cancer, radiation hazards, and diversion of massive healthcare resources.
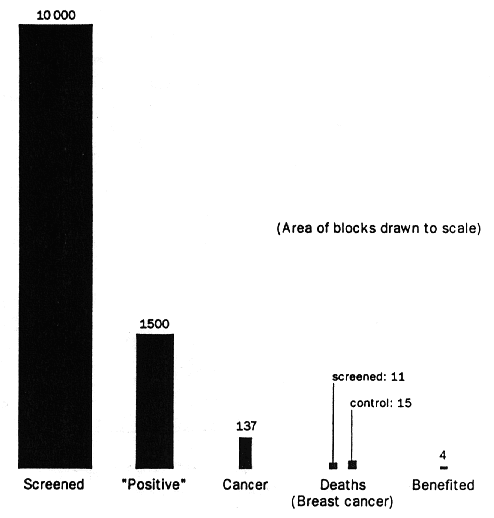
|
|
Figure: Screening mammography: results per
10 000 women after 7 years
Based on SNBH data. |
The most serious concern is the generation of false-positive results.[20] About 1 in 20 women screened have a mammographic abnormality. Between 1 in 5 and 1 in 14 women with a positive screen will be found to have cancer.[21, 22] A positive or suspicious mammogram inevitably leads to further studies or interventions. Stereotactic biopsy and fine-wire needle localisation have become major growth industries. Less invasive diagnostic techniques have improved, but many women still eventually come to open biopsy. Physicians without a surgical background may not realise that operations for non-palpable mammographic "disease" are often more difficult and traumatic than those for a palpable lesion and the results can be unsightly. Fortunately, life-threatening complications of breast surgery are exceedingly rare, but haemorrhage and infections do occur.
The problem of false-positives is compounded by the inclusion of cases of carcinoma in situ in the cancers reported in screening trials. The natural history of carcinoma in situ, whether ductal or lobular, is not well understood but it seems to be a high risk marker rather than an invariable precursor of invasive disease.[23, 24]
The false reassurance of a negative mammogram is another serious issue since 10-15 % of early breast cancers are missed by mammography.[2] There is no justification for the argument that one of the advantages of screening mammography is the comfort and reassurance that cancer is not present.
The financial costs of screening mammography vary widely among different clinical settings and different countries, but the sum must include not only the technical and professional costs of mammography but also all the costs of subsequent interventions and indirect costs to the patient (eg, travel, time off work, home care). The emotional costs, although difficult to quantify, likewise need to be considered.[25] In the Ontario Breast Cancer Screening Program, the cost for each cancer discovered is around $75 000 (£34 800). If a mean figure of population benefit is accepted from the randomised clinical trials (table), around 20 000 women would have to be screened for 1 to benefit. At a low overall estimate of $60.00 (£28) per mammogram, the cost of each woman benefited is $1 2 million (£558 000). This figure accords with a 1988 estimate,[26] and with a detailed cost-benefit analysis by the RAND Corporation in 1995 that reported a range of $166 000 to $1.48 million (£76 000-685 000) for the annual cost per life saved.[27] Further analysis of cost in relation to age-specific mortality would help but would require decades of follow-up data from randomised controlled studies.
Need for a balanced perspective
Screening mammography has been widely publicised, mainly on the basis of the two earliest trials that claim a 30 % reduction in mortality from breast cancer. Little publicity has been given to the results of the four subsequent trials that fail to support the initial studies or to the following facts: (a) the great majority of "positive" screenings are false positives; (b) screening leads to many unnecessary investigations and useless surgery; (c) a "negative" screening result does not mean the absence of breast cancer; and (d) in the large majority of women whose breast cancer is diagnosed by screening the outcome is unchanged. For most women the only "benefit" is extra time spent with the knowledge that they have the disease. It is disappointing that the marginal improvement in terms of reduced mortality is only perceptible in older women. A much greater benefit in life-years gained would be achieved if screening mammography delayed death from breast cancer in younger women, but unfortunately this is not the case.
We fully understand the desperate desire to find something, anything, that might help in this terrible disease. Public imagination has been captured by mammography, and all those involved in the screening industry have a major vested interest. In view of the intense competition for limited resources, those responsible for making allocation/distribution decisions for public healthcare funds must evaluate the evidence more objectively. Although politically attractive, the benefits of mass population screening, even in older women, are too small and the harm and cost generated too great to justify widespread implementation of screening mammography as a publicly funded health measure. To decide otherwise, one must judge that the trauma inflicted by mammography and the resultant surgical interventions in order to benefit 1 woman in 20 000 per year, at a cost in excess of $1 million (£465 000) each, are in the public's best interest.
Our discussion concerns "well women" and the public health policy that should emerge from analysis of the benefits, the costs, and the dangers of screening for breast cancer. The value of mammography for the evaluation of breast disease is not in question here. A patient with breast symptoms or signs has a problem for which she is seeking help, but a well woman with a "positive" mammogram is much more likely to be given a problem where none exists. One can make a case for screening high-risk women -- eg, those with a first-degree family history of premenopausal breast cancer -- because of the high disease prevalence and possibly a higher positive predictive value of mammography in these circumstances. Resources that now go towards mass unselected screening are resources spent on well women and should be channelled into more realistic public education along with casefinding in targeted high-risk groups where the ratio of benefits to harm and the incurred costs may reach a more acceptable level.
References
Mueller CB, Jeffries W. Cancer of the breast: its outcome as measured by the rate of dying and cause of death. Ann Surg 1975; 182: 334-40.
Baker L. Breast cancer detection demonstration project: five-year summary report. Cancer J Clin 1982; 32: 194-225.
Shapiro S. Determining the efficacy of breast cancer screening. Cancer 1989; 63: 1873-80.
Tabar L, Fagerberg CJ, Gad A, Baldetorp L. Reduction in mortality from breast cancer after mass screening with mammography. Lancet 1985; i: 829-32.
Mueller CB. Breast cancer: reporting results with inflationary arithmetic. Am J Clin Oncol 1994; 17: 86-92.
Strax P. Control of breast cancer through mass screening: from research to action. Cancer 1989; 63: 1881-87.
American Cancer Society. Pamphlet 86-(30 mm); no 2077-LE. Washington: ACS, 1986.
Skrabanek R. False premises and false promises of breast cancer screening. Lancet 1985; ii: 316-19.
Schmidt JG. The epidemiology of mass breast cancer screening: a plea for a valid measure of benefit. J Clin Epidemiol 1990; 43: 215-25.
Shapiro S, Venet W, Strax P, Venet L, Roeser R. Ten to fourteen year effects of breast cancer screening on mortality. J Natl Cancer Inst 1982; 69: 349-55.
Andersson I, Aspegren K, Janzon L, et al. Mammographic screening and mortality from breast cancer: the Malmo Mammographic Screening Trial. BMJ 1988; 297: 943-48.
Roberts MM, Alexander FE, Anderson TJ, et al. Edinburgh trial of screening for breast cancer: mortality at 7 years. Lancet 1990; 335: 241 -46.
Frisell J, Eklund G, Hellstrom L, Lidbrink E, Rutqvist LE, Somell A. Randomised study of mammography screening -- preliminary report on mortality in the Stockholm trial. Breast Cancer Res Treat 1991; 18: 49-56.
Miller AB, Baines CJ, To T, Wall C. Canadian National Breast Screening Study: 1. Breast cancer detection and death rates among women aged 40 to 49 years. Can Med Assoc J 1992; 147: 1459-76.
Miller AB, Baines CJ, To T, Wall C. Canadian National Breast Screening Study: 2. Breast cancer detection and death rates among women aged 50 to 59 years. Can Med Assoc J 1992; 147: 1477-88.
Nystrom L, Rutqvist LE, Wall S, et al. Breast cancer screening with mammography: overview of Swedish randomised trials. Lancet 1993 341: 973-78.
Elwood JM, Cox B, Richardson AK. The effectiveness of breast cancer screening by mammography in younger women. Online J Curr Clin Trials [serial online] 25 Feb 1993; (doc no 32): [23,227 words; 195 paragraphs].
Spratt JS, Greenberg RA, Heuser LS. Geometry, growth rates and duration of cancer and carcinoma-in-situ of the breast before detection by screening. Cancer Res 1986; 46: 970-74.
Spratt JA, Von Fournier D, Spratt JS, Weber EE. Mammographic assessment of human breast cancer growth and duration. Cancer 1993 71: 2020-26.
Wright CJ. Breast cancer screening: a different look at the evidence. Surgery 1986; 100: 594-97.
Norton LW, Zeligman BE, Pearlman NW. Accuracy and cost of needle localisation breast biopsy. Arch Surg 1988; 123: 947-50.
Baines CJ, McFarlane DV, Miller AB. Sensitivity and specificity of first screen mammography in 15 NBSS centres. J Can Assoc Radiol 1988; 39: 273-76.
Page DL, Dupont WD, Rogers LW, Landenberger M. Intraductal carcinoma of the breast: follow-up after biopsy only. Cancer 1982; 49: 751-58.
Rosen PP, Kosloff C, Lieberman PH, Adair F, Braun DW. Lobular carcinoma in situ of the breast: detailed analysis of 99 patients with average follow-up of 24 years. Am J Surg Pathol 1978; 2: 225-51.
Marteau T. Psychological costs of screening. BMJ 1989; 299: 527.
Eddy DM, Hasselblad V, McGivney W, Hendee W. The value of mammography screening in women under age 50 years. JAMA 1988; 259: 1512-19.
Kattlove H, Liberati A, Keeler E, Brook R. Benefits and costs of screening and treatment for early breast cancer. JAMA 1995; 273: 142-48.
Department of Health Care and Epidemiology,
University of British Columbia, Vancouver (Prof C J Wright FRCS),
and Department of Surgery, McMastar University, Hamilton,
Ontario, Canada (Prof C B Mueller FRCSC)Correspondence to: Dr C J Wright, Vancouver Hospital and
Health Sciences Centre, 855 West 12th Avenue, Vancouver, BC, V5Z 1M9, Canada