Thank you very much. It’s a pleasure to be here today. A large number of highly radioactive isotopes released by the destruction of the Fukushima Daiichi nuclear power plant grossly contaminated the Japanese mainland. Most of these radionuclides had short half lives which meant they would essentially disappear in a matter of days or months. For many of those who were exposed to them there will be major health consequences.
However, there were some radioactive elements that will not rapidly disappear. And it is these long-lived radionuclides that will remain to negatively affect the health of all complex life forms that are exposed to them.

Chief among them is Cesium-137, which has taken on special significance because it is has proven to be the most abundant of the long-lived radionuclides that has remained in the environment following the nuclear disasters at Chernobyl and Fukushima. It has a 30 year radioactive half life which is why it persists in the environment. Scientists now believe that it will be 180 to 320 years before the Cesium-137 around the destroyed Chernobyl reactor actually disappears from the environment.
Cesium is water soluble and quickly makes its way into soils and waters. It is in the same atomic family as potassium and it mimics it, acting as a macronutrient. It quickly becomes ubiquitous in contaminated ecosystems.
It is distributed by the catastrophic accidents at nuclear power plants because large quantities of volatile radioactive cesium build up inside the fuel rods of nuclear reactors. Thus any accident at a nuclear reactor that causes the fuel rods to rupture, melt, or burn will cause the release of highly radioactive cesium gas.
Long-lived radionuclides such as Cesium-137 are something new to us as a species. They did not exist on Earth in any appreciable quantities during the entire evolution of complex life. Although they are invisible to our senses they are millions of times more poisonous than most of the common poisons we are familiar with. They cause cancer, leukemia, genetic mutations, birth defects, malformations, and abortions at concentrations almost below human recognition and comprehension. They are lethal at the atomic or molecular level.
They emit radiation, invisible forms of matter and energy that we might compare to fire, because radiation burns and destroys human tissue. But unlike the fire of fossil fuels, the nuclear fire that issues forth from radioactive elements cannot be extinguished. It is not a fire that can be scattered or suffocated because it burns at the atomic level—it comes from the disintegration of single atoms.
Thus, radioactivity is a term which indicates how many radioactive atoms are disintegrating in a time period. We measure the intensity of radioactivity by the rate of the disintegrations and the energy they produce.

One Becquerel is equal to one atomic disintegration per second.
One Curie is defined as that amount of any radioactive material that will decay at a rate of 37 billion disintegrations per second.
So one Curie equals 37 billion Becquerels.
Sometimes these man-made radionuclides are compared to naturally occurring radionuclides, such as Potassium-40, which is always found in bananas and other fruits. However this is a false comparison since naturally occurring radioactive elements are very weakly radioactive. In the lab chart the radioactivity is described as the “specific activity”. Note that Potassium-40 has a specific activity of 71 ten millionths of a Curie per gram. Compare that to the 88 Curies per gram for Cesium-137. This is like comparing a stick of dynamite to an atomic bomb.
Highly-radioactive fission products such as Cesium-137 and Strontium-90 emit 10 to 20 million times more radiation per unit volume than does Potassium-40. So which one of these would you rather have in your bananas?
It is in fact the amount of Cesium-137 deposited per square kilometer of land that defines the degree to which an area is classified as being too radioactive to work or live. One may get an idea of the extreme toxicity of Cesium-137 by considering how little of it is required to make a large area of land uninhabitable.
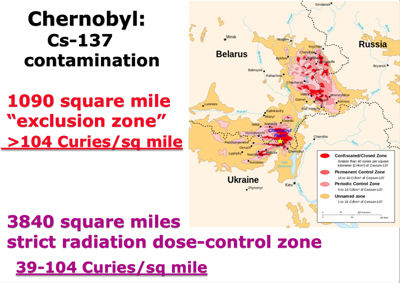
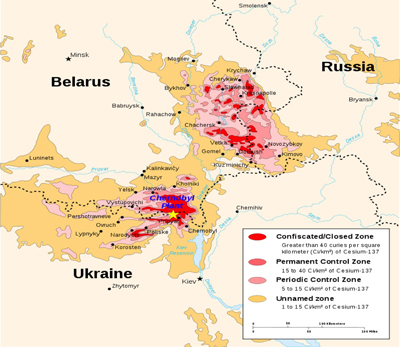
The lands that were grossly contaminated by the destruction of the Chernobyl nuclear power plant are classified by the number of Curies of radiation per square kilometer. There were 3840 square miles of land contaminated with 15 to 40 Curies of radiation per square kilometer. These lands are considered strict radiation dose-control zones.
The 1100 square mile uninhabitable exclusion zone that surrounds the destroyed Chernobyl reactor has greater than 40 Curies of radioactivity per square kilometer. For those more familiar with square miles, that would be 104 Curies per square mile.
Consider again that one gram of Cesium-137 has 88 Curies of radioactivity.
Thus, as little as one third of a gram of Cesium-137, made into microparticles and distributed as a smoke or gas over an area of one square kilometer, will make that square kilometer uninhabitable.
Less than two grams of Cesium-137, a piece smaller than an American dime, if made into microparticles and evenly distributed as a radioactive gas over an area of one square mile, will turn that square mile into an uninhabitable radioactive exclusion zone. Central Park in New York City can be made uninhabitable by 2 grams of microparticles of Cesium-137. Hard to believe, isn’t it?
Remember, these nuclear poisons are lethal at the atomic level. There are as many atoms in one gram of Cesium-137 as there are grains of sand in all the beaches of the world. That’s 1021 atoms—10 to the 21st power. 1480 trillion of them or 1.48 times 10 to the 12th power are disintegrating every second, releasing invisible nuclear energy. So this works out to about one and a half million disintegrations per second per square meter. We can see how this works then.
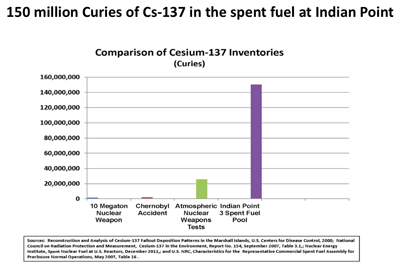
Note the immense inventories of Cesium-137: 150 million Curies that are located in the nearby spent fuel pool at Indian Point Nuclear Power Plant which is about 40 miles from here by road and less than that as the radioactive cloud flies. Many of the 104 US commercial nuclear reactors and power plants have more than 100 million Curies of Cesium-137 in their spent fuel pools. This is many times more than in the spent pools at Fukushima.
So now that we have some idea of the extreme toxicity of Cesium-137, let’s look at the extent of the contamination of the Japanese mainland.

It is now known that the reactors 1, 2, and 3 at Fukushima Daiichi all melted down and melted through the steel reactor vessels within a few days following the earthquake and tsunami of March 11, 2011. This was not made public by either TEPCO or the Japanese government for two months.
The greatest amounts of highly radioactive gases were released shortly after the meltdowns and 80% of this gas released by the reactors is believed to have traveled away from Japan over the Pacific. However the remaining 20% was dispersed over the Japanese mainland.
On March 11th, the US National Nuclear Security Administration offered the use of its NA-42 Aerial Measuring System to the Japanese and US governments. The National Atmospheric Release Advisory Center of the Lawrence Livermore Lab stood up to provide atmospheric modeling projections. The next two slides were produced by Lawrence Livermore and presumably given to the Japanese government.
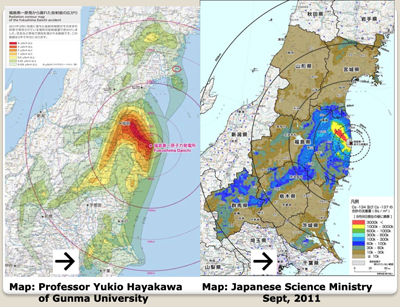
On March 14th, the easterly winds which had been blowing the highly-radioactive gases and aerosols coming from Fukushima out to sea, shifted and pushed the radioactive plume back over the Japanese mainland. You can see the progression. The red indicates the radioactive plume.
Note that the images indicate that the plume first went south over Tokyo and then reversed and went north as the wind changed. All the areas where the radioactive gases passed over were contaminated. However the heaviest contamination occurred where rainfall was occurring and the radiation rained out. This accounts for the patchy deposition of the radioactive fallout.
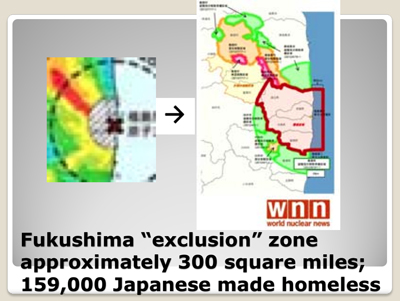
Eight months after the disaster, the Japanese Science Ministry released this map, which shows that 11,580 square miles, which is 30,000 square kilometers, which represents 13% of the Japanese mainland, had been contaminated with long-lived radioactive cesium. Note that the official map does not note any Cesium-137 contamination in the Tokyo metropolitan area, unlike an unofficial survey done at about the same time by Professor Yukio Hayakawa of Gunma University. Given the fact that the Japanese government and TEPCO denied for two months that any meltdowns had occurred at Fukushima, one must look at all official data with a healthy degree of skepticism.
4500 square miles (or earlier today we heard 7700 square miles)—which is an area larger than the size of Connecticut—was found to have radiation levels that exceeded Japan’s previously allowable exposure rate of 1 millisievert per year.
Rather than evacuate this area, Japan chose to raise its acceptable radiation-exposure rate by 20 times, from 1 millisievert to 20 millisieverts per year.
However, approximately 300 square miles adjacent to the destroyed Fukushima reactors were so contaminated that they were declared uninhabitable. 159,000 Japanese were evicted from this radioactive “exclusion zone.” They lost their homes, property, and businesses, and most have received only a small compensation to cover the costs of their living as evacuees.
Note here that the criteria used for evacuation is the millisievert. It is not a measured quantity of radiation per unit area that I have described such as the Curie or Becquerel. Rather the Sievert is a calculated quantity. It’s calculated to represent the biological effects of ionizing radiation. In other words, the millisievert is a derived number, based on the mathematical models which are used to convert the absorbed dose to “effective dose.”
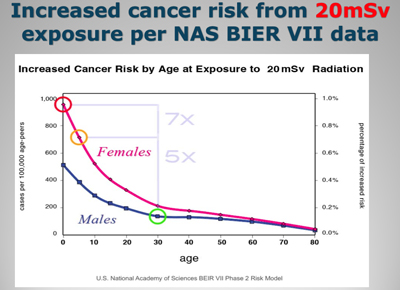
So what is the increased health risk to Japanese based upon their exposure to 20 millisieverts per year? Let us examine figures constructed on the basis of data published by the National Academy of Sciences, courtesy of Ian Goddard.
The vertical Y-axis is calibrated to the number of cancer cases per 100,000 age-peers, and the horizontal X-axis depicts the age of the population, beginning at zero years and moving towards old age. Now examine the allegedly safe dose of 20 millisieverts per year.
As a result of this exposure, there will be about 1000 additional cases of cancer in female infants and 500 cases of cancer in infant boys per 100,000 in their age groups. There will be an additional 100 cases of cancer in 30 year old males per 100,000 in this age peer group.
Notice that children, especially girls, are at the most risk from radiation-induced cancer. In fact a female infant has 7 times greater risk and a 5 year old girl has 5 times greater risk of getting a radiation-induced cancer than does a 30 year old man.
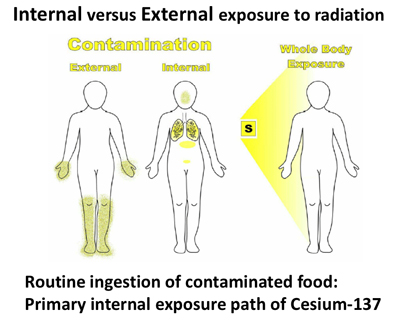
I want to note here that there is a great deal of controversy in regards to the accuracy of the methods used to arrive at the millisievert measurement, especially in regard to an accurate determination of the biological effects of an external versus internal exposure to ionizing radiation.
That is, the effects of an exposure to a source of ionizing radiation that is external to the body, versus an exposure that comes from the ingestion of radionuclides that provide a chronic, long-term internal exposure to living cells, which are adjacent to the radioactive atoms or particles.
In the lands surrounding Chernobyl and Fukushima, the primary route of internal exposure is through the ingestion of foodstuffs contaminated with Cesium-137, which tends to bioaccumulate in plants and animals. What this means is that Cesium-137 cannot be excreted faster than it is being ingested. Thus it accumulates and increases in its concentration in the plant or animal that is routinely ingesting it.
Cesium-137 also tends to biomagnify as it moves up the food chain. This means it becomes progressively more concentrated in predator species. We have seen this before with other industrial toxins, such as DDT, which can magnify its concentration millions of times from the bottom to the top of a food chain.
Consequently, all of the foodstuffs in a contaminated region tend to contain Cesium-137. Those naturally rich in potassium, such as mushrooms and berries, tend to have very high concentrations. Dairy products and meats also tend to have higher concentrations.
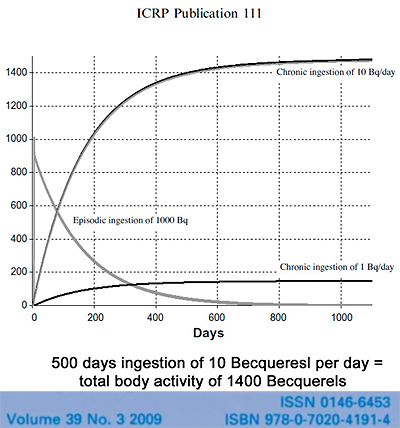
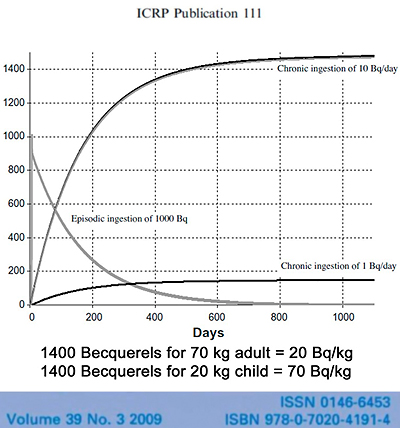
The International Commission on Radiological Protection, the ICRP, which sets radiation safety standards recognizes that Cesium-137 bioaccumulates in humans. This ICRP figure compares a single ingestion of 1000 Becquerels of Cesium-137, a one-time exposure, with the daily ingestion of 10 Becquerels. On the single exposure notice that half the Cesium-137 is gone from the body in 110 days. That’s considered the biological half-life.
Note also that with the routine daily ingestion of 10 Becquerels of Cesium-137 the total radioactivity within the body continues to rise, until after about 500 days there are more than 1400 Becquerels of radioactivity measured in the body. Becquerels can be counted in living persons because the decay of Cesium-137 leads to the emission of gamma radiation which passes through the body and can be measured by a Whole Body Counter. They have a chair that kids, or anyone, can sit and they can calculate the amount of Becquerels per kilogram of body weight.
In a 70 kilogram adult (based on this), a total body activity of 1400 Becquerels would correspond to 20 Becquerels per kilogram of body weight. In a 20 kilogram child it would be 70 Becquerels per kilogram of body weight. The ICRP document does not specify the average age or weight of those examined in the study. However, the safety standards that have been set by the nuclear industry do not consider this level of chronic exposure to so-called “low-dose” radiation to be a significant danger to human health.
The ICRP states in this document that a whole body activity of 1400 Becquerels is equivalent to an exposure of one-tenth a millisievert per year. In other words, the radiation models used by radiation biologists that convert this level of internal absorbed dose to “effective dose,” do not predict serious health risks from such exposures. In fact they state that it is safe to have 10 times this exposure level.
There is however strong evidence that the ingestion of these levels of so-called “low-dose” radiation are, in fact, particularly injurious to children. Research done by Dr. Yuri Bandazhevsky, and his colleagues and students, in Belarus during the period 1991 through 1999, correlated whole body radiation levels of 10 to 30 Becquerels per kilogram of whole body weight with abnormal heart rhythms and levels of 50 Becquerels per kilogram of body weight with irreversible damage to the tissues of the heart and other vital organs.
One of the key discoveries made by Bandazhevsky was that Cesium-137 bioconcentrates in the endocrine and heart tissues, as well as the pancreas, kidneys and intestines. This goes completely against one of the primary assumptions used by the ICRP to calculate “effective dose” as measured by milliseiverts: that Cesium-137 is uniformly distributed in human tissues.
Let me restate that. The current ICRP methodology is to assume that the absorbed dose is uniformly distributed in human tissues. This is, in fact, not the case.
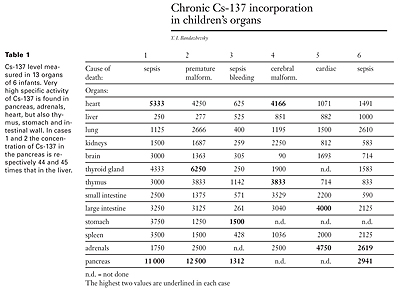
This table, taken from Bandazhevsky’s “Chronic Cs-137 incorporation in children’s organs,” compares the radioactivity measured in 13 organs of 6 infants. Very high specific activity, that is, levels of radioactivity, often 10 times higher than in other organs and tissues were found in the pancreas, thyroid, adrenal glands, heart, and intestinal walls.
Bandazhevsky summarized his nine years of research in his study entitled, “Radioactive Cesium and the Heart” [2001]. With the help of friends I have just finished editing a new Russian-to-English translation of this work [2013].
It was never previously translated in large part because shortly after Dr. Bandazhevsky presented it to the Parliament and the President of Belarus, he was summarily arrested and imprisoned. Government agents entered the Medical Institute which he directed and destroyed his archived slides and samples. After he was released from prison he was held under house arrest. It was during this time that he actually wrote the study. He did so in an attempt to preserve his research knowing that he was about to be imprisoned again for a very long time.
Just as Soviet physicians were forbidden to diagnose a radiation-related illness following Chernobyl, the Belarusian government acted to suppress the work of Bandazhevsky who had been protesting government efforts to resettle people back into land badly contaminated with Cesium-137.
In “Radioactive Cesium and the Heart,” Bandazhevsky also did a correlation between the amount of Cesium-137 in live children and their heart function. He worked with the BELRAD Institute, which conducted more than 100,000 Whole Body Counts on Belarusian children, measuring the amounts of internally ingested Cesium-137 in each child.
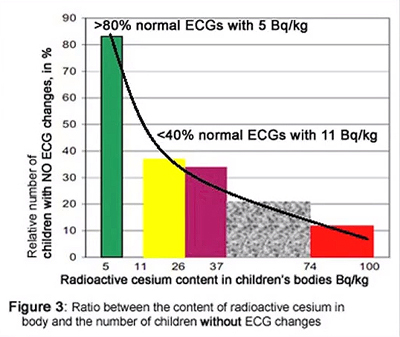
There were so many contaminated children in Belarus that it was difficult to find any with zero Becquerels per kilogram. However only those with less than 10 Becquerels per kilogram of body weight had normal Electro Cardiograms [ECGs]. 35% of the children with 11 to 37 Becquerels per kilogram had normal ECGs. 20% of children with 37 to 74 Becquerels per kilogram had normal ECGs. And only 11% of those with 74 to 100 Becquerels per kilogram had normal ECGs.
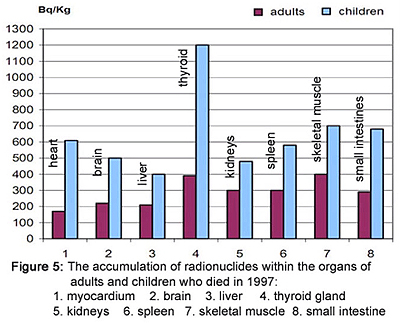
This slide, which shows the averaged results from hundreds of autopsies done during 1997, is also taken from “Radioactive Cesium and the Heart.” Notice the very high concentration of Cesium-137 in the thyroid gland.
While we generally worry about radioactive iodine concentrating in the thyroid, Bandazhevsky’s work shows us that Cesium-137 is likely to play a major role in thyroid cancer too.
I want to point out again that the currently accepted medical and legal understanding of Cesium-137 is that it is “distributed fairly uniformly” in human tissues. I copied the web page from the US EPA website, from which this quote is taken. Clearly, the autopsied human tissue samples analyzed by Bandazhevsky show that this is not the case. This new understanding needs to be incorporated into the way we understand how internally ingested radionuclides act upon the human body.
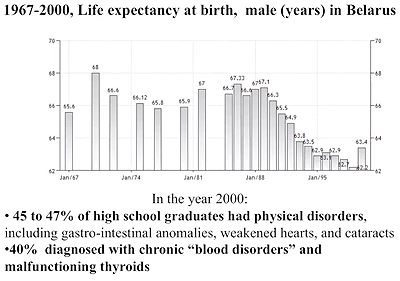
Two million people in Belarus live on lands severely contaminated by Cesium-137. Most of the children that live there are not considered to be healthy although they were before the nuclear power plant at Chernobyl exploded in 1986. Fourteen years after the explosion, 45 to 47 percent of high school graduates had physical disorders, including gastro-intestinal anomalies, weakened hearts, and cataracts. And 40% were diagnosed with chronic “blood disorders” and malfunctioning thyroids.
I am afraid that there are many Japanese people now living on lands equally contaminated with radioactive cesium. If Japanese children are allowed to routinely ingest foodstuffs contaminated with Cesium-137, they will likely develop the same health problems that we see now in the children and teenagers of Belarus and Ukraine.
Thus it is very important that we recognize the danger posed to children by the routine ingestion of contaminated food with Cesium-137 where ever they might live. It is also important to prevent further nuclear disasters which release these fiendishly toxic poisons into the global ecosystems. Given the immense amounts of long-lived radionuclides which exist at every nuclear power plant this is an urgent task.

I hope I have made it clear that long-lived radionuclides produced by nuclear power plants are neither “safe” nor “clean.” I would suggest that it is very bad idea to manufacture these nuclear poisons to try to make electricity, that it is past time we stopped manufacturing them and try to manage those which we have already created which must be isolated from the ecosystems for at least 100,000 years.
Thank you.